Probiotics applications in bone cancer treatment
What are Probiotics?
The term “probiotic”, as defined by the World Health Organization (WHO), refers to strains of live bacteria that, when ingested in adequate amounts, provide health benefits to users (WHO, 2002). In other words, probiotics or beneficial microorganisms, when used, help balance the digestive system, enhance health, and prevent diseases.
The current probiotic strains commonly used belong to three groups: Lactobacillus, Bifidobacterium, and yeast (Saccharomyces) (Pradhan et al., 2020). Among these, Lactobacillus encompasses over 100 different species, primarily acid-producing bacteria. This strain is often found in the human and animal digestive systems. Bifidobacterium (such as Bacillus subtilis and Bacillus clausii) has the capability to produce spores that can survive in stomachs with low pH conditions. Therefore, it is extensively used in pharmaceuticals and food. Some yeast species are also being researched and evaluated for their safety and effectiveness as probiotics.
Currently, probiotics or beneficial microorganisms are synthesized, packaged in high concentrations, properly formulated, and easily stored. Moreover, their use is generally safe as they do not cause adverse effects and aid in improving health, especially when using harmful medications affecting the digestive and immune systems. Particularly for cancer patients undergoing therapies like chemotherapy and radiation, resulting in side effects such as diarrhea, oral ulcers, fever, or fatigue, the use of probiotics can minimize these side effects during treatment, thereby enhancing quality of life.
Role of Probiotics in Bone Cancer Treatment
Bone cancer originates from bone cells. Cancer cells can either start within the bone (primary) or spread to the bone from cells in other parts of the body (secondary). In the first case, cancer may arise from bone-forming cells, cartilage cells, or connective tissue cells within the bone. Primary bone cancer is rare, accounting for only 1% of diagnosed cancers, usually occurring in adolescents and young adults aged 15-25 (Seigel et al., 2018).
Types of bone cancer can be categorized based on the types of bone cells that give rise to cancer: Osteosarcoma (from bone-forming cells), Chondrosarcoma (from cartilage cells), Ewing sarcoma (from supportive or connective tissue), and Chordoma (from remnants of the spine) (Singh et al., 2015). The treatment for bone cancer depends on the type, size, location, and condition of the tumor, as well as the age and health of the patient. Commonly used methods include surgery, chemotherapy, radiation therapy, cryosurgery (using liquid nitrogen to freeze and kill cancer cells), and targeted therapy. Chemotherapy and radiation therapy are often combined with surgical procedures for their effectiveness in destroying bone cancer cells (Chen et al., 2017). However, up to 45% of patients experience side effects from these treatments, such as anemia, diarrhea, constipation, and oral ulcers, affecting their quality of life and sometimes necessitating treatment interruptions due to health issues. Hence, probiotics are used as a method to reduce and improve the adverse effects of chemotherapy and radiation therapy, aiming to enhance immunity, prevent metastasis, or inhibit cancer cell growth.
Certain strains of Lactobacillus probiotics have been shown to be effective in reducing gastrointestinal side effects related to chemotherapy and radiation therapy, such as diarrhea, abdominal pain, vomiting, and bloating. Over 90% of studies show that patients experience symptom reduction when using probiotics. These microorganisms help balance the gut microbiota, enhance gut immunity, and thereby improve quality of life (Rodriguez et al., 2021). Strains like B. longum, L. lactis, and E. faecium have demonstrated the reduction of mucosal inflammation and oral ulcers in patients undergoing radiation therapy within a timeframe of 1 to 7 weeks of usage (Jiang et al., 2019).
Additionally, probiotics can be used to regulate the process of cancer cell self-destruction (Apoptosis). They reduce the expression of cancer-causing genes and the transformation of DNA within cells, thereby inhibiting the growth, metastasis, and preventing (Figure 1) (Sankarapandian et al., 2022) based on metabiotics (components, structures, metabolites, signaling molecules of probiotic strains that help regulate physiological activities within the host cells, Shenderov et al., 2018). Sharma et al.’s 2011 study showed that probiotics play a role in regulating cell apoptosis by modulating the expression of Bcl-2 and Bax surface proteins. This helps protect cells from self-destruction, reducing the risk of cancer. Wang et al.’s 2019 research indicated that surface proteins of Lactobacillus acidophilus strains can activate the self-destruction ability (autophagy) of cancer cells by triggering oxidative molecules, paving the way for using probiotics in cancer treatment.
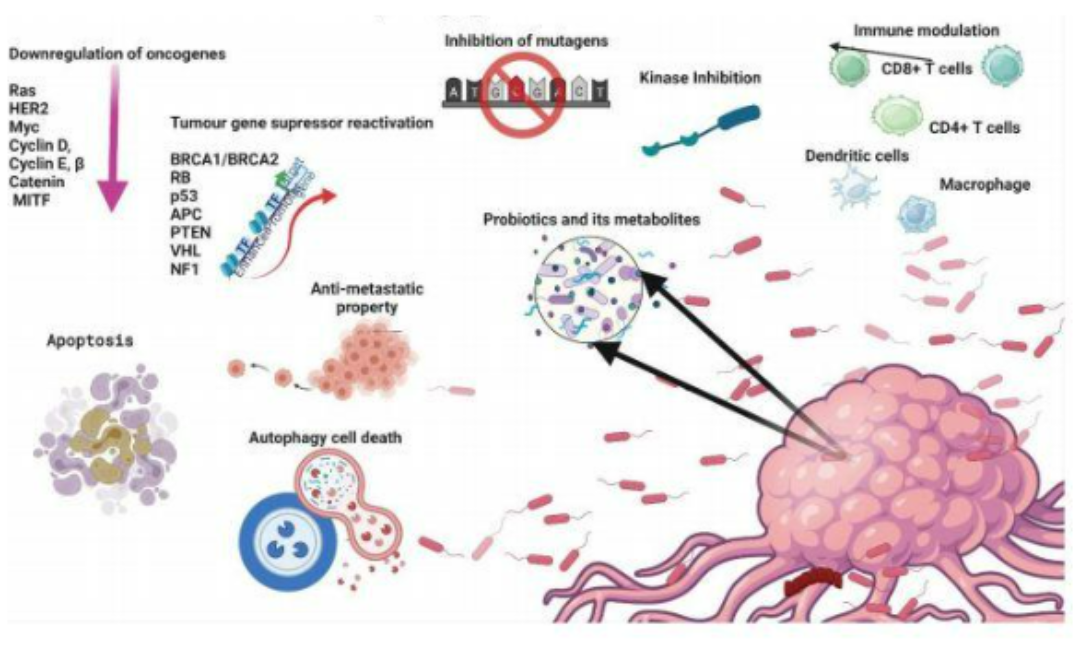
Figure 1: Description of the role of probiotics in cancer treatment and prevention (apoptosis process, immune enhancement, DNA mutations, cancer gene expression, cell self-destruction) (Sankarapandian et al., 2022)
In conclusion, probiotics have proven to be effective in adjunct therapy for patients undergoing chemotherapy and radiation therapy. Moreover, there is increasing evidence and studies demonstrating the role of probiotics in cancer treatment and prevention by regulating immunity and reducing cancer gene expression. Therefore, the potential for using probiotics in cancer treatment and prevention is significant, although further trials are needed to evaluate their effectiveness and safety.
References
- FAO/WHO. “Food and Agriculture Organization of the United Nations/World Health Organization. Guidelines for the Evaluation of Probiotics in Food.” (2002).
- Pradhan, Diwas, Rashmi H. Mallappa, and Sunita Grover. “Comprehensive approaches for assessing the safety of probiotic bacteria.” Food Control108 (2020): 106872.
- Siegel, Rebecca L., Kimberly D. Miller, and Ahmedin Jemal. “Cancer statistics, 2018.” CA: a cancer journal for clinicians68, no. 1 (2018): 7-30.
- Singh, Tejinder, Veerpal Kaur, Manish Kumar, Prabhjot Kaur, R. S. R. Murthy, and Ravindra K. Rawal. “The critical role of bisphosphonates to target bone cancer metastasis: an overview.” Journal of Drug Targeting23, no. 1 (2015): 1-15.
- Chen, Clark, John Garlich, Katie Vincent, and Earl Brien. “Postoperative complications with cryotherapy in bone tumors.” Journal of bone oncology7 (2017): 13-17.
- Rodriguez-Arrastia, Miguel, Adrian Martinez-Ortigosa, Lola Rueda-Ruzafa, Ana Folch Ayora, and Carmen Ropero-Padilla. “Probiotic Supplements on Oncology Patients’ Treatment-Related Side Effects: A Systematic Review of Randomized Controlled Trials.” International journal of environmental research and public health18, no. 8 (2021): 4265.
- Jiang, Chunling, Huan Wang, Chaofei Xia, Qing Dong, En Chen, Yang Qiu, Yong Su et al. “A randomized, double‐blind, placebo‐controlled trial of probiotics to reduce the severity of oral mucositis induced by chemoradiotherapy for patients with nasopharyngeal carcinoma.” Cancer125, no. 7 (2019): 1081-1090.
- Shenderov, Boris A. “Metabiotics: novel idea or natural development of probiotic conception.” Microbial ecology in Health and Disease24, no. 1 (2013): 20399.
- Sharma, S., R. L. Singh, and P. Kakkar. “Modulation of Bax/Bcl-2 and caspases by probiotics during acetaminophen induced apoptosis in primary hepatocytes.” Food and chemical toxicology49, no. 4 (2011): 770-779.
- Sankarapandian, Vidya, Balu Alagar Venmathi Maran, Ramya Lakshmi Rajendran, Manasi P. Jogalekar, Sridharan Gurunagarajan, Rajapandiyan Krishnamoorthy, Prakash Gangadaran, and Byeong-Cheol Ahn. “An Update on the Effectiveness of Probiotics in the Prevention and Treatment of Cancer.” Life12, no. 1 (2022): 59.
- Wang, Huifang, Xian Cheng, Li Zhang, Shichen Xu, Qiuxiang Zhang, and Rongrong Lu. “A surface-layer protein from Lactobacillus acidophilus NCFM induces autophagic death in HCT116 cells requiring ROS-mediated modulation of mTOR and JNK signaling pathways.” Food & function10, no. 7 (2019): 4102-4112.
