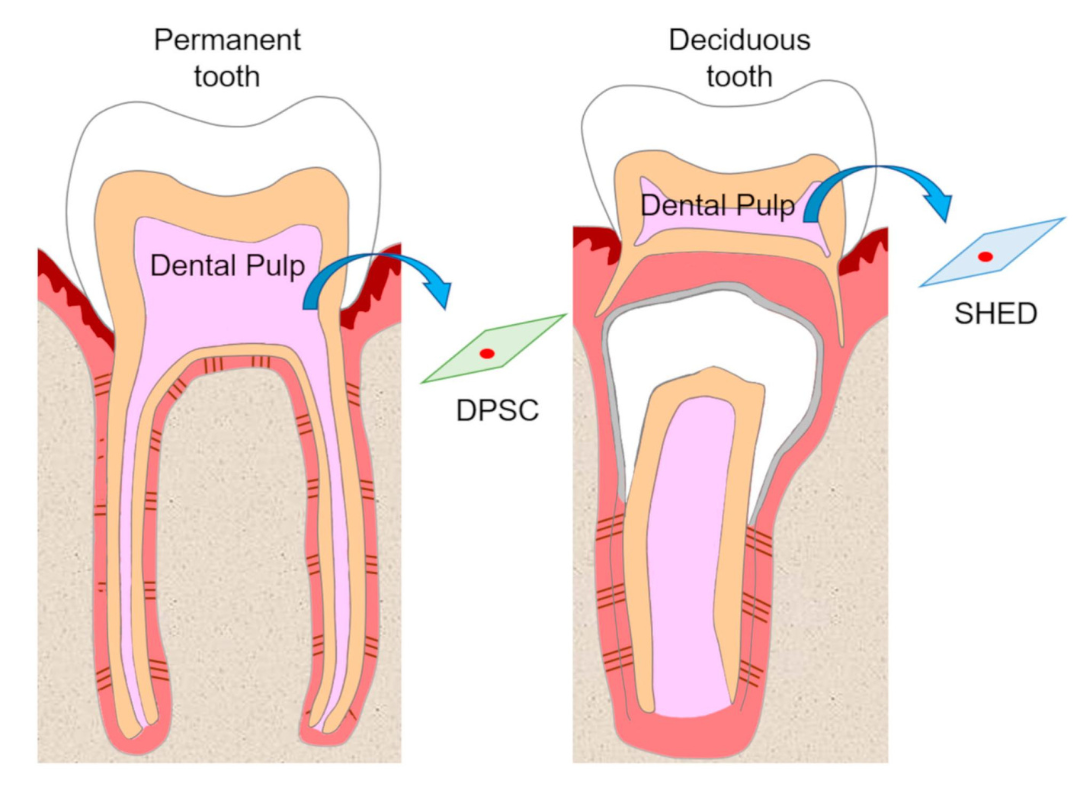
Similarities and Differences between Dental Pulp and Umbilical Cord Stem Cells
Studies to date have shown that mesenchymal stem cells (MSCs) are an attractive source of tissue engineering and regenerative medicine due to their self-renewal capacity and multi-lineage differentiation potential [1]. Although bone marrow and adipose tissue are the major sources of scientific research and clinical therapy, they have certain limitations, including low proliferation and differentiation capabilities, invasive sampling procedures, and limited widespread applicability [2,3]. Therefore, to overcome these limitations, it is necessary to conduct further research and development of alternative MSC sources. Recently, umbilical cord (UC) and dental pulp (DP) MSCs have garnered much attention because of their convenient collection process, excellent proliferative and differentiation abilities, low susceptibility to bacterial and viral infections, and ethical considerations in medical practice.
Similarities of Dental Pulp and Umbilical Cord Stem Cells
Several potential therapeutic applications of these MSCs in various disease models have been reported, such as neurodegenerative disorders, low-grade inflammation, post-stroke paralysis, and diabetes [4,5,6,7]. However, there have been few studies on the biological characteristics, including morphology, proliferation capacity, apoptosis resistance, multi-lineage differentiation potential, and immunophenotype of UC- and DP-MSCs to select the appropriate MSC source for future clinical applications.
Characteristic antigens on dental pulp and umbilical cord stem cells: flow cytometry analysis showed that all MSCs were negative for specific hematopoietic or endothelial cell antigens CD14, CD34, and CD45, regardless of early or late stages, with surface antigen expression rates <5%. However, they were positive for the expression of specific mesenchymal markers CD29, CD44, and CD90, with surface antigen expression rates >95%. Both cell types maintained cell surface antigen expression and survival ability after 10 days of culture [3].
Differences between Dental Pulp and Umbilical Cord Stem Cells
In 2016, Ren et al. compared the biological characteristics of MSCs derived from dental pulp and umbilical cord [3]. The results showed that the proliferation capacity of UC-MSCs appeared to be superior to DP-MSCs, but the rates of senescence and apoptosis were less observed in DP-MSCs at each stage. Importantly, MSCs gradually lost their proliferation capacity but tended to undergo senescence and apoptosis along with long-term culture, consistent with previous studies [8].
These phenomena may be partly explained by the switch from glycolysis to oxidative phosphorylation energy metabolism [9]. Although more energy can be generated to support cell proliferation through oxidative glucose metabolism in mitochondria (36 mol adenosine triphosphate (ATP) per mol glucose) compared to glycolysis (only 2 mol ATP per mol glucose in the conversion of glucose to pyruvate and then lactate), multiple reactive oxygen species (ROS) reactions also occur, ultimately contributing to oxidative stress, cellular aging, and programmed cell death [10]. Therefore, like tumors and malignant cells, MSC maintenance mainly relies on glycolysis rather than mitochondrial glucose metabolism to generate ATP even in the presence of oxygen (a phenomenon known as the Warburg effect) [11].
UC-MSCs showed a higher ability to produce lactate compared to the other two MSC sources from dental pulp and menstrual blood. When assessing the morphology of the two cell types on adherent culture dish surfaces, it was observed that MSCs from umbilical cord had a characteristic polygonal shape and included cell pellet clusters. DP-MSCs maintained a good fibroblast-like morphology. These findings suggest that DP-MSCs may better maintain the morphology of stem cells after long-term culture.
Differentiation Ability between Dental Pulp and Umbilical Cord Stem Cells
Regarding the differentiation potential of the two cell types, a study used MSCs from both sources cultured in osteogenic and adipogenic induction media. The osteogenic process was determined by calcium deposition, while the adipogenic process was determined by the formation of lipid droplets. The results showed a successful differentiation between osteoblasts and adipocytes. Quantitative analysis and comparison demonstrated the adipogenic potential between DP- and UC-MSCs, but the osteogenic process of DP-MSCs was significantly better than that of UC-MSCs even at the late stage [3].
The proliferation rates of both MSC sources have also been evaluated. The growth curves showed slow cell proliferation in the first two days, followed by logarithmic growth, continuing for about 4-5 days, and then reaching a plateau phase in cell growth on the 6th-7th day. UC-MSCs appeared to proliferate significantly faster than DP-MSCs on the second day. The cell proliferation time of UC-MSCs was found to be significantly shorter than that of DP-MSCs. Therefore, UC-MSCs showed the highest proliferation, followed by DP-MSCs [3].
This was further demonstrated by analyzing cell viability stained with Alamar Blue and metabolic activity assessed by glucose consumption and lactate production. Interestingly, there were minimal differences in cell quantity, cell viability, and glucose/lactate levels among the three different stages, indicating that high proliferation capacity was maintained even after 10 culture cycles.
Senescence of Dental Pulp and Umbilical Cord Stem Cells
To determine whether the senescence capability of the cells is similar in UC- and DP-MSCs, β-galactosidase staining was performed. The results showed that the cell senescence process in DP-MSCs was significantly slower than that in UC-MSCs at the second and tenth passages, but there was no significant difference in cell senescence between DP- and UC-MSCs at the sixth passage.
In addition, flow cytometry analysis also showed that the apoptotic process in both cell types was low, with DP-MSCs having approximately 10 times lower apoptosis rates compared to UC-MSCs. At the tenth passage, there was no significant difference in apoptosis rates between DP- and UC-MSCs. Therefore, DP-MSCs demonstrated better resistance to apoptosis, followed by UC-MSCs.
Human umbilical cord and dental pulp mesenchymal stem cells possess self-renewal and multipotent capabilities. Compared to UC-MSCs, DP-MSCs may have advantages in the treatment of bone injuries due to their higher osteogenic differentiation potential and lower rates of apoptosis, senescence, and cell proliferation at an appropriate level. However, researchers still need to conduct further in-depth comparative experiments to evaluate their potential in neural, cardiac, hepatic, pancreatic, or other lineage differentiations and investigate their therapeutic efficacy in the body.
Refferences:
[1] Hilfiker A., Kasper C., Hass R., Haverich A. Mesenchymal stem cells and progenitor cells in connective tissue engineering and regenerative medicine: is there a future for transplantation? Langenbeck’s Archives of Surgery. 2011;396(4):489–497.
[2] Li Y., Charif N., Mainard D., Bensoussan D., Stoltz J.-F., de Isla N. Donor’s age dependent proliferation decrease of human bone marrow mesenchymal stem cells is linked to diminished clonogenicity. Bio-Medical Materials and Engineering. 2014;24(1, supplement):47–52.
[3] REN, Huaijuan, et al. Comparative analysis of human mesenchymal stem cells from umbilical cord, dental pulp, and menstrual blood as sources for cell therapy. Stem cells international, 2016, 2016.
[4] Wu L. W., Wang Y.-L., Christensen J. M., et al. Donor age negatively affects the immunoregulatory properties of both adipose and bone marrow derived mesenchymal stem cells. Transplant Immunology. 2014;30(4):122–127.
[5] Rezai Rad M., Wise G. E., Brooks H., Flanagan M. B., Yao S. Activation of proliferation and differentiation of dental follicle stem cells (DFSCs) by heat stress. Cell Proliferation. 2013;46(1):58–66.
[6] Liu Y., Mu R., Wang S., et al. Therapeutic potential of human umbilical cord mesenchymal stem cells in the treatment of rheumatoid arthritis. Arthritis Research & Therapy. 2010;12(6, article R210)
[7] Hu J., Yu X., Wang Z., et al. Long term effects of the implantation of Wharton’s jelly-derived mesenchymal stem cells from the umbilical cord for newly-onset type 1 diabetes mellitus. Endocrine Journal. 2013;60(3):347–357.
[8] Sethe S., Scutt A., Stolzing A. Aging of mesenchymal stem cells. Ageing Research Reviews. 2006;5(1):91–116.
[9] Pattappa G., Thorpe S. D., Jegard N. C., Heywood H. K., de Bruijn J. D., Lee D. A. Continuous and uninterrupted oxygen tension influences the colony formation and oxidative metabolism of human mesenchymal stem cells. Tissue Engineering C: Methods. 2013;19(1):68–79.
[10] Fu W., Li J., Chen G., Li Q., Tang X., Zhang C. Mesenchymal stem cells derived from peripheral blood retain their pluripotency but undergo senescence during long-term culture. Tissue Engineering—Part C: Methods. 2015;21(10):1088–1097.
[11] Zhang L., Marsboom G., Glick D., et al. Bioenergetic shifts during transitions between stem cell states (2013 Grover Conference series) Pulmonary Circulation. 2014;4(3):387 – 394
