Stem cell therapy in post-stroke treatment and recovery
Inadequate blood supply causes damage to the brain in a condition known as a stroke, also referred to as a cerebrovascular accident. Blood clots form in the brain, interrupting blood flow and causing blockages in arteries, leading to ruptured blood vessels and subsequent bleeding in the brain. When arteries burst, there’s insufficient oxygen and nutrients supplied to the brain’s nerve cells, causing them to die if not immediately addressed. Stroke survivors often suffer severe neurological impairments, such as partial paralysis, speech loss, behavioral disorders, and emotional changes (1).
Stroke ranks as the second leading cause of death globally, affecting around 12.2 million people and causing 6.5 million deaths each year (2). Approximately 87% of strokes occur due to local blood insufficiency. The incidence of stroke increases with age, doubling for those over 55 years old. However, the incidence of strokes in people aged 20 to 54 has increased from 12.9% to 28.6% between 1990 and 2016 (3). Stroke patients typically suffer nerve damage, and therefore, methods aiding neurological recovery in stroke patients are the primary research focus. Stem cell use, especially mesenchymal stem cells (MSCs), is a new trend because of the crucial properties of stem cells in regeneration and repair. Among these, using mesenchymal stem cell therapy is leading in stem cell therapies for stroke patients (3).
Stroke and its characteristics
Blood supply to the brain comes from two sources: the internal carotid arteries arising from the neck and the vertebral arteries (forming the Circle of Willis). The internal carotid arteries branch to create two major arteries: the anterior cerebral artery and the middle cerebral artery. The right and left vertebral arteries meet at the brainstem to form the basilar artery on the belly surface of the brainstem, constituting the posterior cerebral circulation (4). Nerve cells are highly sensitive to oxygen and glucose levels. If the blood supply is damaged or insufficient, someone needs to restore it promptly to prevent temporary or permanent damage to nerve cells. Stroke is classified into two types: ischemic stroke, accounting for 80% caused by blood clots or thrombi blocking blood vessels, and 20% as hemorrhagic stroke due to blood vessel rupture (5,6). Stroke complications usually relate to nerve damage, such as sleep disorders, partial or full body paralysis, loss of sensory functions like walking, vision, perception, sensation, memory, and decreased cognitive function (6).
Current treatment strategies for stroke patients involve using drugs to protect and reduce damage to the nervous system when ischemia occurs, such as Clomethiazole (reducing glutamate-induced toxicity), Mexiletine, and venous thrombolytic drugs that improve blood transportation. However, these methods are only effective in cases intervened within 9 or 24 hours after a stroke (7,8). The effectiveness of current methods in restoring function after a stroke is quite limited. As a result, most stroke survivors suffer lifelong nerve damage due to incomplete reconstruction and the function of the damaged brain tissue (9). Finding novel approaches to developing effective stroke treatments for both acute and post-stroke phases is a crucial direction in research today. Stem cell therapy represents a promising, safe, and effective avenue for stroke patients.
Role and mechanism of mesenchymal stem cells in stroke treatment
Mesenchymal stem cells (MSCs), defined by the International Society for Cellular Therapy (ISCT), must meet specific criteria for human MSCs for both scientific research and clinical application. They should adhere to plastic surfaces in culture flasks (adherent cells), fully express the surface markers Cluster of Differentiation (CD) CD73, CD90, and CD10 while not expressing CD11b, CD14, CD19, CD34, CD45, CD79 α, HLA-DR when examined using flow cytometry, and have the ability to differentiate into bone, cartilage, and fat cells in experimental conditions (10). MSCs have an unlimited proliferation capacity for culture conditions and undergo genetic stability assessments to avoid cell lines with chromosomal abnormalities. MSCs usually exhibit good immune tolerance, expressing low levels of Major Histocompatibility Complex class I (MHC I) and little or no expression of MHC II, not stimulating T and NK cells. Therefore, allogeneic MSC transplantation is safe and avoids unwanted adverse effects (11). The mechanism of MSCs in treating stroke patients is still under research and not entirely explained, but some mechanisms that help improve the condition of stroke patients have been identified (Figure 1).
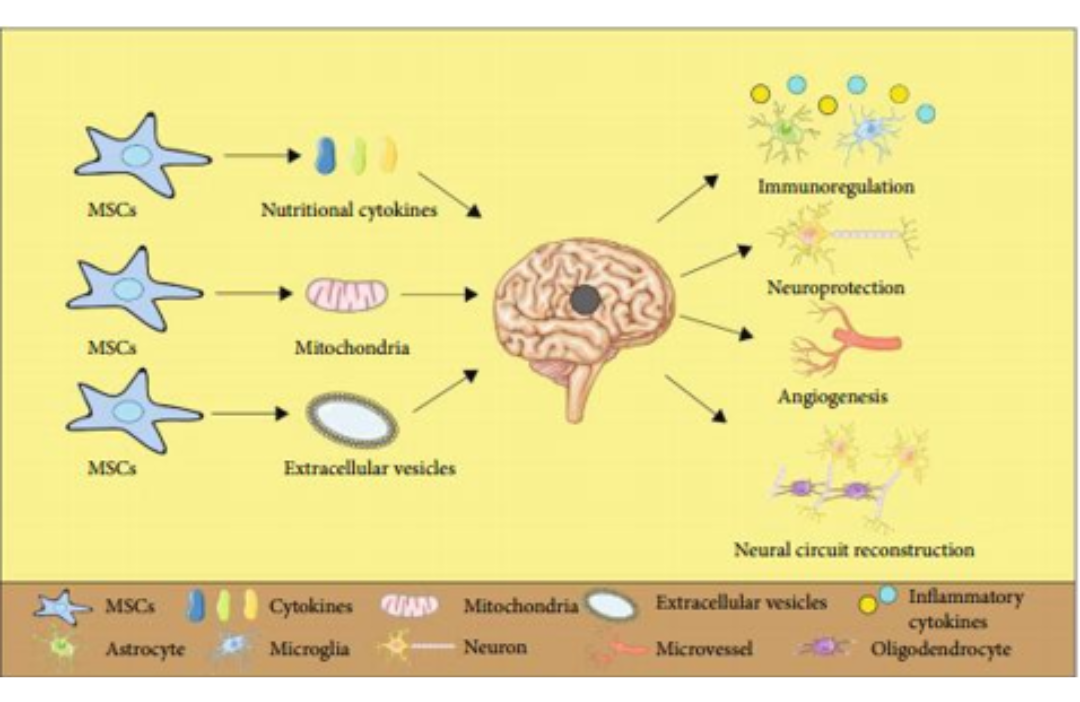
Figure 1: Mechamisms of MSC in stroke treatment
MSCs can modulate the immune response and inflammation. After a stroke, damaged nerve cells and brain tissue release oxidizing agents and pro-inflammatory cytokines that stimulate inflammatory cells, attracting numerous inflammatory factors that destroy the blood-brain barrier. Continuous invasion of immune cells can lead to nerve cell necrosis, brain edema, and increased secondary brain damage in stroke patients (12). Studies show that MSCs have the ability to release anti-inflammatory cytokines like TGF-β, preventing immune cell migration to the area of stroke (13). Huang’s research combined MSCs with oxygen and glucose-deprived nerve cells and showed that MSCs have anti-inflammatory capabilities by secreting IL-6 and reducing the expression of pro-inflammatory cytokines like TNF-α (14).
MSCs can protect nerve cells during a stroke. Local blood insufficiency in a stroke often causes a core area of local blood deprivation in the center and a penumbra of local blood deprivation surrounding it. Therefore, protecting the non-dead nerve cells around the core area is crucial in stroke treatment (15). MSCs show potential in preventing programmed cell death of nerve cells. Zhu’s study transplanted MSCs into rabbits suffering from localized blood deprivation stroke and found that the group treated with MSCs showed increased expression of the protein Bcl-2, which prevents programmed cell death. Simultaneously, there was a decrease in the expression of protein p53, a type of protein associated with nerve cell death by the programmed cell death pathway. MSCs can also release nerve growth factors such as basic fibroblast growth factor (bFGF), brain-derived neurotrophic factor (BDNF), and nerve growth factor (NGF) (16-18).
MSCs have the ability to form blood vessels. After a stroke, the blood vessels in the brain become damaged, causing injury to the blood-brain barrier. MSCs can secrete substances such as vascular endothelial growth factor (VEGF) to promote the formation of immature blood vessels and angiopoietin-1 (Ang-1) involved in the maturation and stabilization of vessels (19,20). The potential for MSCs to differentiate into nerve cells and supporting nerve cells is the goal of MSC transplantation in stroke treatment. However, experiments injecting MSCs into the brain’s outer layer surrounding the area affected by stroke have shown signs of immaturely differentiated nerve cells. Yet, they establish necessary ion channels to generate active potentials (21). Hence, MSC differentiation into nerve cells isn’t a mechanism for stroke treatment, but they have the capability to promote migration and survival of nerve progenitor cells in the surrounding area of local ischemia (22). Other studies show MSCs can promote the reorganization of nerve connections by inhibiting nerve fiber inhibitory proteins and increasing synaptophysin expression (23). The transplantation of MSCs or their extracellular vesicles (EVs) derived from MSCs, which are cell-free components secreted by MSCs, has also shown efficacy in stroke treatment.
Clinical trials using MSCs in stroke treatment
Trials using MSCs in stroke treatment have shown promising results in both preclinical studies using MSC stem cells for stroke treatment and animal model experiments. Thus, researchers have implemented and evaluated the application of MSC stem cells for patient treatment to assess its effectiveness. A study conducted by Bang and colleagues involved clinical research on 30 patients with stroke because of local cerebral ischemia, of whom only 5 received autologous MSC transplantation with a dose of 1×108 cells via intravenous injection after 5-7 weeks, while the remaining group did not receive MSCs. During the one-year observation period, no adverse effects were observed in the patients, and improvements in neurological deficits were continuously recorded in both groups. The results indicated that in the group receiving MSCs, monitoring indices such as Barthel (evaluating daily activities) and the mRS scale (Modified Rankin Scale: assessing stroke patient self-care abilities) consistently improved at 3, 6, and 12 months of observation, with no observed adverse effects or abnormal changes in cell and hematologic evaluations (24).
Another clinical trial involving 85 patients aged 30-75 with severe local cerebral ischemia due to stroke was conducted by Lee and colleagues over a 5-year period. Patients were randomly divided into two groups: those treated with autologous MSCs at a dosage of 1×108 cells/patient via intravenous infusion and those not using MSCs (control group). The assessment results showed that the number of patients with mRS scores from 0 to 3 (0 – no symptoms at all, 3 – moderate symptoms but able to walk independently) in the MSC-treated patients significantly increased, while the control group’s scores decreased. No serious adverse effects were observed in either study group (25).
A Phase I/II clinical trial conducted by Levy and colleagues on chronic stroke patients with significant disabilities due to stroke involved administering allogeneic MSCs via intravenous infusion at dosages ranging from 0.5, 1, and 1.5×106 cells/kg body weight in Phase 1 and 1.5×106 cells/kg body weight in Phase 2. Results from both phases showed that the Barthel index increased from 6.8 ± 11 to 10.8 ± 15.5 after 12 months of treatment. The percentage of patients with Barthel scores >95 points (completely independent) increased from the initial 11.4% to 27.6% after 8 months and 33.5% after 12 months. These results indicate that intravenous infusion and using allogeneic MSCs in patients with local cerebral ischemia due to stroke are safe and improve daily functional activities (26). Deng and colleagues are currently conducting another Phase II clinical trial to evaluate the safety and effectiveness of infusing allogeneic bone marrow-derived MSCs (BM-MSCs) into patients with local cerebral ischemia due to stroke for the first time. A group of 59 patients received BM-MSC infusion four times a week at a dose of 1×106 cells/kg body weight via intravenous infusion. The mRS scale was used to evaluate treatment effectiveness, and no adverse effects were observed after 90 days. The researchers are still conducting the trial, and they have not reported specific results yet (27). Additionally, the Steinberg research group conducted a study involving the transplantation of SB623 cells, a line of BM-MSCs genetically modified with Notch-1, into the brains of 18 patients with chronic stroke. The results showed significant improvements in the European Stroke Scale (ESS), NIHSS score, total Fugl-Meyer score (FM), and FM exercise score after 24 months of treatment (28).
The application of MSCs in stroke treatment offers hope for improving and recovering from post-stroke injuries. Further in-depth research on the mechanisms and roles of MSCs in recovering from stroke-induced injuries helps in better understanding and devising appropriate treatment strategies for patients, as well as establishing a robust foundation for clinical transitions in MSC-based therapies.
REFERENCES:
- Kuriakose, D., & Xiao, Z. (2020). Pathophysiology and treatment of stroke: present status and future perspectives. International journal of molecular sciences, 21(20), 7609.
- Feigin, V. L., Brainin, M., Norrving, B., Martins, S., Sacco, R. L., Hacke, W., … & Lindsay, P. (2022). World Stroke Organization (WSO): global stroke fact sheet 2022. International Journal of Stroke, 17(1), 18-29.
- Collaborators, G. B. D. S. (2019). Global, regional, and national burden of stroke, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol, 18(5), 439-458.
- Purves D, Augustine GJ, Fitzpatrick D, et al., editors. Neuroscience. 2nd edition. Sunderland (MA): Sinauer Associates; 2001. The Blood Supply of the Brain and Spinal Cord. Available from: https://www.ncbi.nlm.nih.gov/books/NBK11042/
- Musuka, T. D., Wilton, S. B., Traboulsi, M., & Hill, M. D. (2015). Diagnosis and management of acute ischemic stroke: speed is critical. Cmaj, 187(12), 887-893.
- Powers, W. J., Rabinstein, A. A., Ackerson, T., Adeoye, O. M., Bambakidis, N. C., Becker, K., … & American Heart Association Stroke Council. (2019). Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke, 50(12), e344-e418.
- Campbell, B. C., Ma, H., Ringleb, P. A., Parsons, M. W., Churilov, L., Bendszus, M., … & Hsu, J. P. (2019). Extending thrombolysis to 4· 5–9 h and wake-up stroke using perfusion imaging: a systematic review and meta-analysis of individual patient data. The Lancet, 394(10193), 139-147.
- Nogueira, R. G., Jadhav, A. P., Haussen, D. C., Bonafe, A., Budzik, R. F., Bhuva, P., … & Jovin, T. G. (2018). Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. New England Journal of Medicine, 378(1), 11-21.
- Grefkes, C., & Fink, G. R. (2020). Recovery from stroke: current concepts and future perspectives. Neurological research and practice, 2(1), 1-10.
- Dominici, M. L. B. K., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F. C., Krause, D. S., … & Horwitz, E. M. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy, 8(4), 315-317.
- Chen, C. (2014). From mesenchymal stem cell therapy to discovery of drug therapy for systemic sclerosis (Doctoral dissertation, University of Southern California).
- Shichita, T., Ito, M., & Yoshimura, A. (2014). Post-ischemic inflammation regulates neural damage and protection. Frontiers in cellular neuroscience, 8, 319.
- Yoo, S. W., Chang, D. Y., Lee, H. S., Kim, G. H., Park, J. S., Ryu, B. Y., … & Suh-Kim, H. (2013). Immune following suppression mesenchymal stem cell transplantation in the ischemic brain is mediated by TGF-β. Neurobiology of disease, 58, 249-257.
- Huang, P., Gebhart, N., Richelson, E., Brott, T. G., Meschia, J. F., & Zubair, A. C. (2014). Mechanism of mesenchymal stem cell–induced neuron recovery and anti-inflammation. Cytotherapy, 16(10), 1336-1344.
- Dabrowska, S., Andrzejewska, A., Lukomska, B., & Janowski, M. (2019). Neuroinflammation as a target for treatment of stroke using mesenchymal stem cells and extracellular vesicles. Journal of Neuroinflammation, 16(1), 1-17.
- Zhu, Y., Guan, Y. M., Huang, H. L., & Wang, Q. S. (2014). Human umbilical cord blood mesenchymal stem cell transplantation suppresses inflammatory responses and neuronal apoptosis during early stage of focal cerebral ischemia in rabbits. Acta Pharmacologica Sinica, 35(5), 585-591.
- Nomura, T., Honmou, O., Harada, K., Houkin, K., Hamada, H., & Kocsis, J. D. (2005). I.V. infusion of brain-derived neurotrophic factor gene-modified human mesenchymal stem cells protects against injury in a cerebral ischemia model in adult rat. Neuroscience, 136(1), 161–169.
- Chen, J., Li, Y., Katakowski, M., Chen, X., Wang, L., Lu, D., … & Chopp, M. (2003). Intravenous bone marrow stromal cell therapy reduces apoptosis and promotes endogenous cell proliferation after stroke in female rat. Journal of neuroscience research, 73(6), 778-786.
- Zhang, Y., Ma, L., Su, Y., Su, L., Lan, X., Wu, D., … & Ji, X. (2019). Hypoxia conditioning enhances neuroprotective effects of aged human bone marrow mesenchymal stem cell-derived conditioned medium against cerebral ischemia in vitro. Brain Research, 1725, 146432.
- Toyama, K., Honmou, O., Harada, K., Suzuki, J., Houkin, K., Hamada, H., & Kocsis, J. D. (2009). Therapeutic benefits of angiogenetic gene-modified human mesenchymal stem cells after cerebral ischemia. Experimental neurology, 216(1), 47–55.
- Zhao, L. R., Duan, W. M., Reyes, M., Keene, C. D., Verfaillie, C. M., & Low, W. C. (2002). Human bone marrow stem cells exhibit neural phenotypes and ameliorate neurological deficits after grafting into the ischemic brain of rats. Experimental neurology, 174(1), 11-20.
- Song, M., Mohamad, O., Gu, X., Wei, L., & Yu, S. P. (2013). Restoration of intracortical and thalamocortical circuits after transplantation of bone marrow mesenchymal stem cells into the ischemic brain of mice. Cell transplantation, 22(11), 2001-2015.
- Shen, L. H., Li, Y., Chen, J., Cui, Y., Zhang, C., Kapke, A., … & Chopp, M. (2007). One-year follow-up after bone marrow stromal cell treatment in middle-aged female rats with stroke. Stroke, 38(7), 2150-2156.
- Bang, O. Y., Lee, J. S., Lee, P. H., & Lee, G. (2005). Autologous mesenchymal stem cell transplantation in stroke patients. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society, 57(6), 874-882.
- Lee, J. S., Hong, J. M., Moon, G. J., Lee, P. H., Ahn, Y. H., & Bang, O. Y. (2010). A long-term follow-up study of intravenous autologous mesenchymal stem cell transplantation in patients with ischemic stroke. Stem cells, 28(6), 1099-1106.
- Levy, M. L., Crawford, J. R., Dib, N., Verkh, L., Tankovich, N., & Cramer, S. C. (2019). Phase I/II study of safety and preliminary efficacy of intravenous allogeneic mesenchymal stem cells in chronic stroke. Stroke, 50(10), 2835-2841.
- Deng, L., Peng, Q., Wang, H., Pan, J., Zhou, Y., Pan, K., … & Wang, Y. (2019). Intrathecal injection of allogenic bone marrow-derived mesenchymal stromal cells in treatment of patients with severe ischemic stroke: study protocol for a randomized controlled observer-blinded trial. Translational Stroke Research, 10, 170-177.
- Steinberg, G. K., Kondziolka, D., Wechsler, L. R., Lunsford, L. D., Kim, A. S., Johnson, J. N., … & Schwartz, N. E. (2018). Two-year safety and clinical outcomes in chronic ischemic stroke patients after implantation of modified bone marrow–derived mesenchymal stem cells (SB623): a phase 1/2a study. Journal of Neurosurgery, 131(5), 1462-1472.
